Many studies have shown that plant-based diets and Mediterranean diets can lower the risk of development of gestational diabetes mellitus. Plants have been the main source of medicines since ancient times. Despite tremendous advances in medicinal chemistry, synthetic drugs have not provided cures to many diseases due to their adverse side effects or diminution in response after prolonged use. Medicinal mushrooms have been used traditionally as an anti-diabetic food for centuries especially in countries such as China, Japan, India and Korea. These are source of natural bioactive compounds. The bioactive constituents are polysaccharides, proteins, dietary fibres, lectins, lactones, alkaloids, terpenoids, sterols and phenolic compounds which have various health benefits. This review will focus on recent examples of diverse types of mushrooms that have been validated by scientific evaluation as having promising activity for the prevention and/or treatment of gestational diabetes mellitus. Dietary components and plant-derived molecules can be used in the future to complement current treatment strategies for gestational diabetes mellitus.
1.
Introduction
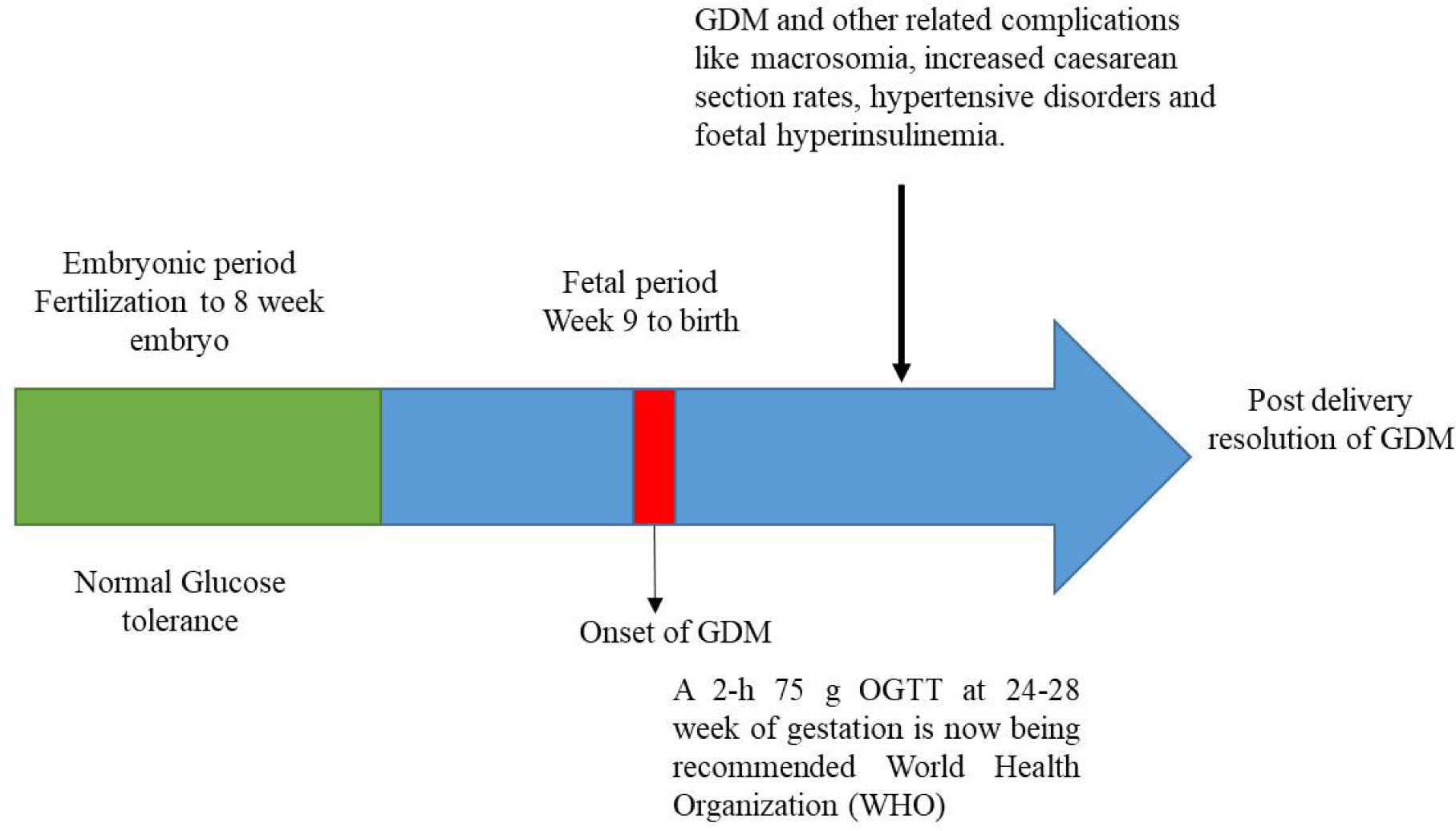
Gestational diabetes is a form of hyperglycemia. It is a common metabolic disorder during pregnancy. According to ‘World Health Organization’ (WHO), gestational diabetes is defined as hyperglycaemia that is first recognised during pregnancy and is observed to precedes to type 2 diabetes in up to one third of women later in life [1]. Series of endocrine, metabolic and vascular changes take place during pregnancy to fulfil the demand of energy and nutrient needs of the foetus. GDM develops when pancreatic β-cell is not sufficient to compensate for the decreased insulin sensitivity and due to which there is increase in post-prandial free fatty acids, hepatic glucose production, insulin resistance and glucose production [2]. Although, glucose homeostasis is restored after delivery but there is high risk of development of type 2 diabetes mellitus in future. Gestational diabetes indicates the underlying frequency of type 2 diabetes for any ethnic and population group with incidences rising globally needing huge health care and economic costs [3].
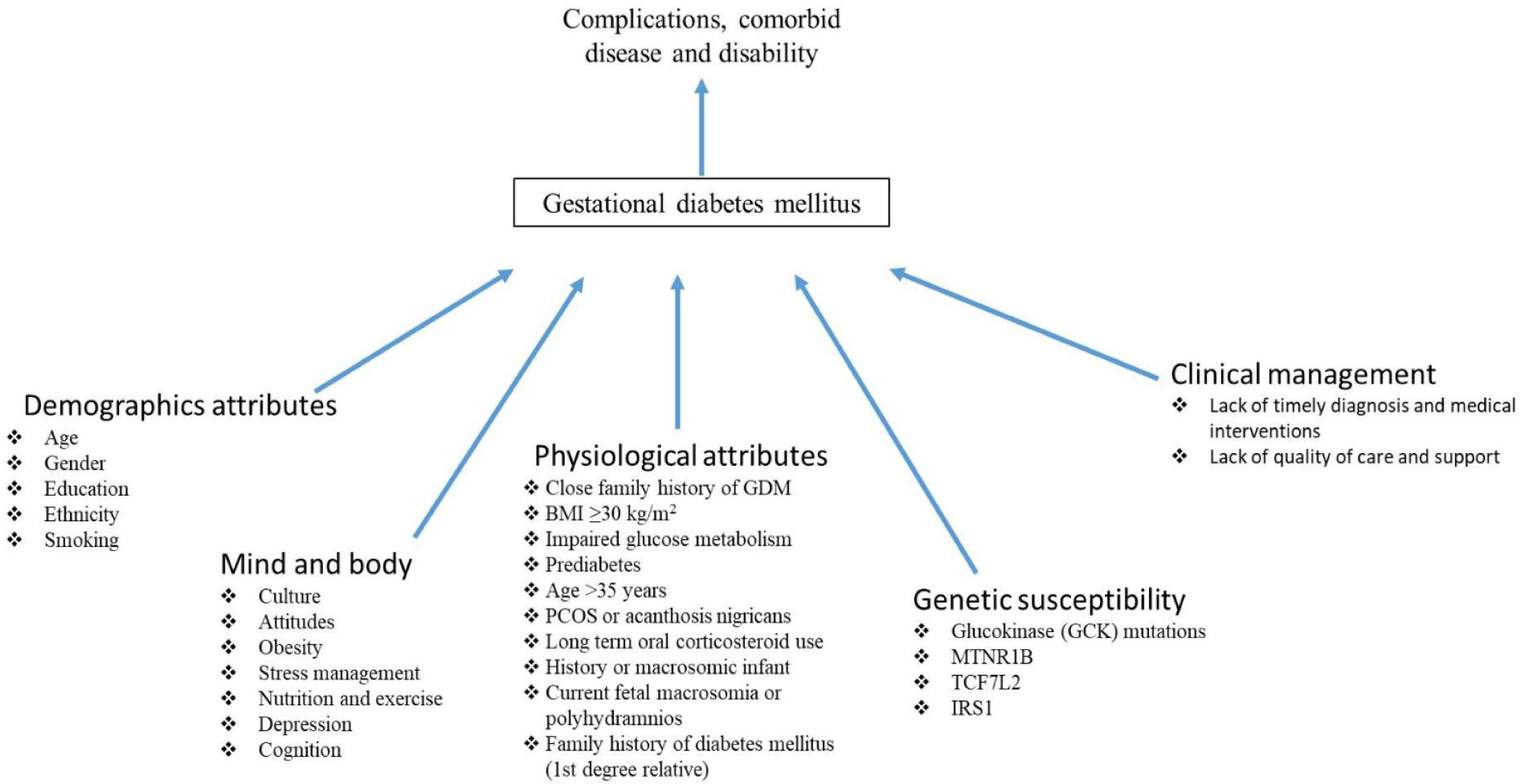
GDM is clearly a major public health issue internationally, especially among those of non-European descent and data from previous studies also revealed that GDM was more prevalent in women with family history of diabetes, women who were obese and parous [4].
2.
Pathophysiology
During pregnancy, there is an increase in oestrogen and progestin concentration which further leads to decrease fasting glucose, delay in gastric emptying and increase in appetite. Tissue sensitivity to insulin decreases as gestation progresses and therefore, post-prandial glucose concentration increases. To counteract this, pancreatic β-cell of the mother have to increase insulin secretion to regulate glucose concentrations and GDM females are unable to produce sufficient insulin (Figure 1). Insulin resistance is also caused by defects in receptors of insulin -signalling cascade. The down-regulation of insulin receptor substrate-1 (IRS-1) decreases insulin-mediated glucose uptake in skeletal muscles. Genetic variations have also been associated with greater risk of developing gestational diabetes (Figure 2) [5].
3.
Management of gestational diabetes mellitus
Blood glucose monitoring is very important to manage GDM. Diet and nutrition must be carefully monitored, and exercise can effectively regulate the blood glucose concentrations and can reduce the risks of complications for a mother as well as her baby due to hyperglycaemia [5]. Maintaining healthy body weight, abstinence from cigarette smoking, restricting refined sugars and Trans fats, consumption of low glycaemic foods, soluble fibres and calorie restriction are the preventive measures in blood glucose regulation [6].
Plants have been used historically for medicinal purposes worldwide. Plants are the rich sources of fibres, vitamins, minerals and phytonutrients and have shown beneficial effects in management of diabetes, cardiovascular disorders, obesity and some cancers [7]. In recent years, mushrooms and seaweeds have gained an interest of many researchers and now they have moved on to explore mushrooms to control and manage post-prandial hyperglycemia [8]. Mushrooms have been used in Traditional Chinese Medicine (TCM) and as functional foods in nutraceuticals for their anti-diabetic and anti-obesity effects [9]. The worldwide mushroom industry has grown, and rate of consumption has been increased at a rapid rate since the late 1990s [10].
Mushrooms have been part of culinary culture across the globe and have been valued for their unique flavor. More than 2000 mushroom species exist in nature but only 25 species have been accepted as food [11]. Eighty five percent of the world's mushroom supply is from five main genera such as Agaricus (A. bisporus and A. brasilensis) contributing 30% world's cultivated mushrooms. Pleurotus, second one with 5 to 6 cultivated species, constitutes about 27% of world's output and then Lentinula edodes (Shiitake) contributes 17%, whereas Auricularia and Flammulina constitute for only 6% and 5% respectively [10].
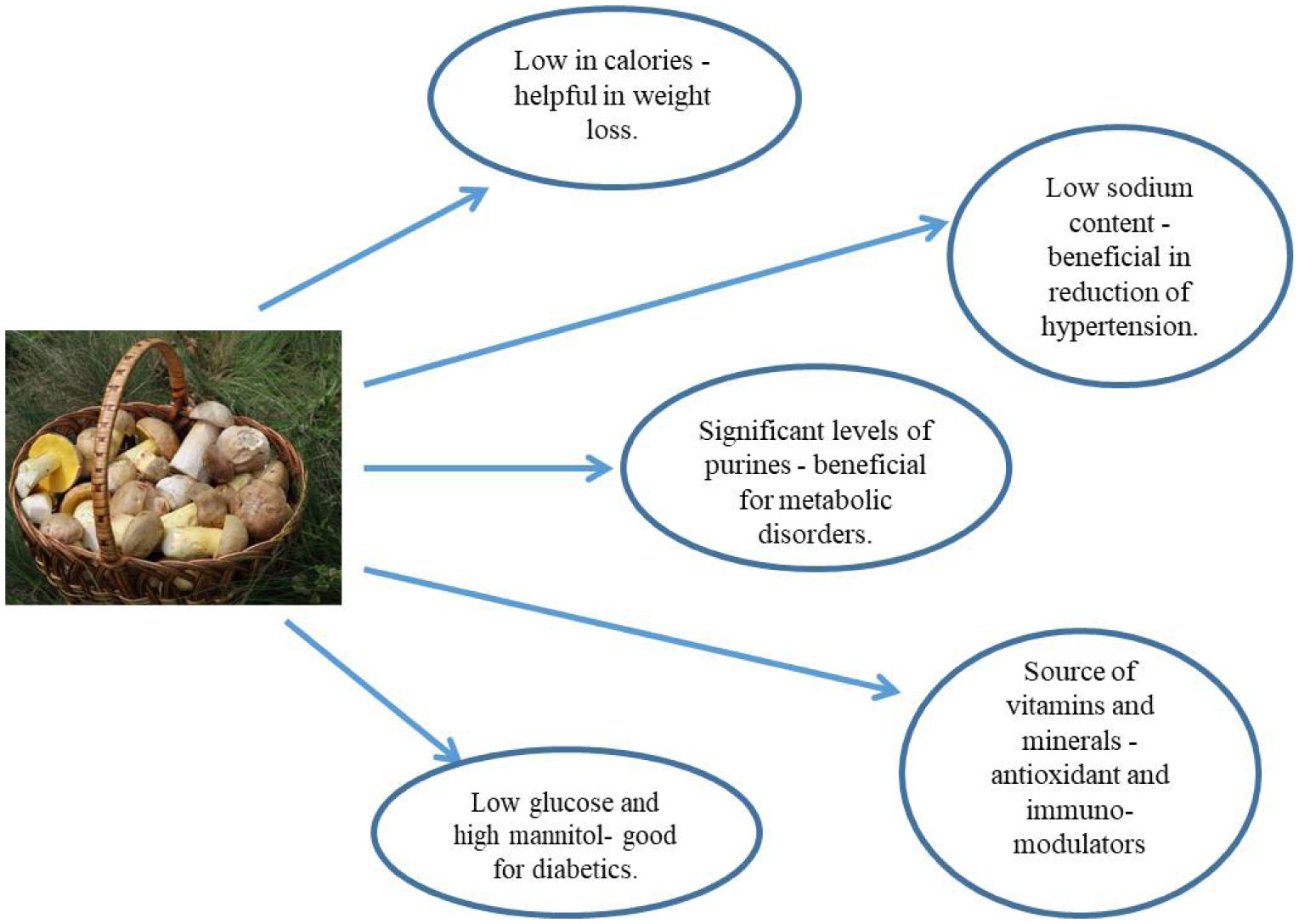
Mushrooms belong to phylum Basidiomycota and order, Agaricales. They have a fruiting body composed of a stipe (stalk), pileus (cap) and lamellae (gills) and mycelium, the root absorbs the nutrients [12]. Gymnopilus edulis seaweed is potential glucosidase enzyme inhibitor, Lenitus edodes, Ganoderma lucidum have shown to possess anti-diabetic effects in various studies [13]. Several beneficial nutritional effects of mushrooms have been identified (Figure 3) [14].
The aim of this article is to review the relevant literature on use of different mushrooms in managing diabetes and gestational diabetes mellitus.
4.
Medicinal mushrooms with potential anti-diabetic activities
4.1. Ganoderma lucidum (Lingzhi/Reishi)
Ganoderma lucidum belongs to the family Ganodermaceae which is widely used for longevity and health promotion. It is commonly known as ‘Ligzhi’ in China and ‘Reishi’ in Japan. The bioactive compounds are polysaccharides, β-glucans, lectins, organic germanium, phenols, steroids, amino acids, lignins, mycins and vitamins. Ganoderma is considered as golden medicinal fungus [15]. Ganoderma lucidum is often combined with another mushroom, Cordyceps sinensis which is thought to increase potency of Ganoderma lucidum [16].
4.2. Lentinus edodes (Shiitake mushrooms)
Shiitake mushrooms have been used to treat common cold for hundreds of years. These mushrooms contain high fibre, high levels of β-glucans, proteins, lipids, carbohydrates, minerals, vitamin B1, B2 and C, ergosterol, lectins and lentinans. Studies have shown that shiitake mushrooms could alleviate the damage of pancreatic β-cells, promote insulin synthesis and lower plasma glucose levels. An ex-polymer of L. edodes reduced plasma glucose and increased insulin synthesis in streptozotocin-induced diabetic rats compared to normal rats [8].
4.3. Ophiocordyceps sinensis
Ophiocordyceps earlier known as Cordyceps sinensis has been used historically as a medicinal mushroom. It has only been found growing at high altitudes in Qinghai-Tibetan plateau and due to high demand and declining harvest, this has become the one of the most expensive mushrooms used in oriental medicine. This mushroom is only found in co-habitation with larvae of an insect, needs rarefied atmosphere, mineral rich soil and low temperature which makes artificial cultivation challenging. Cordyceps has a unique profile of secondary metabolites and thus, considered as world's most costly medicinal mushroom [17]. Various animal and human studies have shown antihyperglycemic, anti-atherosclerotic, anti-hypertensive, antioxidant and cholesterol lowering effects of Cordyceps sinensis [16].
4.4. Agaricus blazei Murill
ABM is native to Brazil and widely grown in Japan. This mushroom is traditionally believed to fight physical and emotional stress, stimulate immune system, manage diabetes, cholesterol, digestive issues. Ergosterols, lignins and polysaccharides are the main bioactive constituents [18]. The polysaccharides, α-glucans, β-glucans have been shown to possess immune-modulatory and anti-mutagenic activity in both in-vitro as well as in-vivo studies [8]. The natural polysaccharides of this mushroom have shown to be effective in regulating diabetes by the mechanisms given below [19].
Elevation of plasma insulin and decrease of pancreatic glucagon
Increase in insulin sensitivity and improvement of insulin resistance
Inhibition of α-glucosidase enzyme
Increase of hepatic glycogen and inhibition of sugar dysplasia
Increased use of glucose from peripheral tissues
Scavenging free radicals and lipid peroxidation
4.5. Grifola frondosa
Grifola frondosa, also known as Maitake mushrooms has varied medicinal properties and is popular in Korea, China and Japan. Chinese people have been consuming Maitake mushrooms for hundreds of years for it's enticing taste. These mushrooms possess anti-tumour, immune-regulatory, anti-diabetic, beneficial effects against hepatitis and HIV/AIDS [20].
4.6. Pleurotus pulmonarius (Grey Oyster Mushroom)
This type of mushroom is an excellent anti-diabetic food. It contains polysaccharides, vitamins and minerals, dietary fibres and rich in essential amino acids. Oyster mushroom is consumed all over the world due to its high nutritive value and flavour. It can be cultivated within wide-range of temperatures and possess wide range of health benefits [21].
4.7.
Antrodia cinnanomea
This mushroom is native to Taiwan with various medicinal properties. This has mainly used to treat diarrhoea, abdominal pain, hypertension, cancer and toxicities caused by various foods and drugs [11].
4.8.
Panellus serotinus (Mukitake)
This mushroom is the most delicious edible mushroom in Japan. This mushroom has found be very beneficial for liver and helps to prevent the development of non-alcoholic fatty liver disease [11].
4.9.
Auricularia auricular
Auricularia species are widely cultivated in China, Taiwan, Thailand, Philippines, Indonesia, Malaysia [10]. Most species of Auricularia are edible and possess medicinal properties. A. polytricha (Woodear mushrooms) and A. auricula-judae (black fungus) are anti-tumour, anti-coagulant and cholesterol lowering, A.auricula-judae has also shown hypoglycemic properties [11].
4.10.
Flammulina velutipes
This mushroom is traditionally used for soups in China. Bioactive constituents are dietary fibre and polysaccharides. Flammulina is hypoglycemic, cholesterol lowering and anti-oxidant [11].
5.
Hypoglycemic effects of mushrooms
There have been numerous investigations conducted on rats to understand effect of different variety of medicinal mushrooms on underlying biomarkers of diabetes; some of these studies are summarized in Table 1. All these studies have either used polysaccharides, protein complexes, or compounds extracted from fruiting bodies, cultured mycelium, or cultured broth of medicinal mushrooms. This section includes most of the medicinal mushrooms that have been investigated for their potential in Diabetes along with the demonstrated mechanism of action. The medicinal mushroom extracts are invaluable source of a variety of:
-
enzymes such as laccase, superoxide dismutase, glucose oxidase, peroxidase;
-
polysaccharides such as β-D-glucans, glucuronoxylomannan;
-
lectins, glucoproteins, proteins, polysaccharide-protein complexes,
-
lipopolysaccharides,
-
terpenoids, alkaloids, sterols, phenolic substances, metal chelating agents [19].
These components have been demonstrated to target various deranged carbohydrate metabolic pathways thereby lowering blood glucose and insulin concentrations in type 2 diabetics as shown in Table 2 and 1. Efforts are currently underway to quantify these compounds for their therapeutic usage in diabetes and other conditions.
One of such studies experimented with the mycelia powder extracted from Agaricus brasiliensis and Ganoderma lucidum. The extract was fed to streptozotocin (STZ)-induced diabetic Wistar outbred white male rats. The study demonstrated hypoglycaemic effects when compared to control group as evidenced by lowered blood glucose and glycosylated hemoglobin concentrations [22]. This study also demonstrated that when the mushroom preparations were orally administered at a dose of 1 g/kg for 14 days to diabetic rats, the physiological balance between sialylation and desialylation processes was restored and integrity of erythrocyte membrane surface glycoconjugates strengthened. Thus, mushrooms extract prevented dysregulation of iron homeostasis erythrocyte membranes thereby strengthening them and causing hypoglycemic and anti-anemic action [22]. Another animal experiment tested the polysaccharides and α-glucans extracted from the body of Grifola Frondosa and reported decreased concentrations of fasting plasma glucose, triglycerides, cholesterol and increased serum insulin in diabetic mouse [20]. Another study utilized Maitake mushrooms (Grifola frondosa) and reported lowered blood glucose concentration. The fall in blood glucose may be attributed to Vanadium and an alpha-glucose inhibitor present in this variety of mushrooms. [8]. Vanadium complexes have been demonstrated to have insulin-mimetic as well as insulin potentiation properties [59] as observed in in vivo, animal and human studies. It facilitates regulation of glucose metabolism in diabetics as it plays a pivotal role being a cofactor for enzymes that participate in insulin signalling leading to GLUT4 translocation [60]–[62].
Further, a study reported reduction in hyperglycaemia induced oxidative-nitrative stress on rats with gestational diabetes [gestational diabetes was induced by streptozotocin (80 mg/kg), administered intraperitoneally] when medicinal mushroom powder was orally administered at a dose of 1 g/kg body weight [26]. Another experiment of daily dose of 100 or 200 mg/kg of lyophilized and reconstituted Lentinus edodes to diabetic rats on 1st to the 19th day of gestation, observed reduced plasma catalase, glucose, urea, triglycerides, and aspartate aminotransferase [23].
Ingestion of mycelia, broth, and mycelia plus broth of Maitake (Grifola frondosa) has also been demonstrated to alleviate the increase in 2 h postprandial blood glucose. The experiments showed significant improvement in innate immunity by modulating levels of inflammatory cytokines such as tumor-necrosis factor-α, macrophages productions of IFN-γ from the T-leukocytes, IL-4, and IL-6 from the monocytes and IL-4 from the T-splenocytes type 2 diabetes mellitus rats [24].
Pleurotus ostreatus and P. cystidiosus ingestion has also been shown to reduce blood glucose level, improved serum lipid profile, liver glycogen, liver function enzymes, and enzymic antioxidants activities in pancreas, liver, and kidney in STZ-induced diabetic rats [27]. Another mushroom Phellinus rimosus has been shown to increase antioxidant status in pancreas, liver, and kidney tissues with concomitant decrease in levels of thiobarbituric acid- reactive substances thus showcasing significant hypoglycemic and hypolipidemic activities that may be related to its insulinogenic and antioxidant effect of medicinal mushrooms [25].
Studies by Huang et al. [63],[64] demonstrated that polysaccharides from Pleurotus tuber-regium mushrooms have antihyperglycemic properties and they reduce oxidative stress in diabetic rats that were fed a high fat diet.
Another interesting compound-Ternatin (a highly methylated cyclic heptapeptide) isolated from mushroom Coriolus versicolor was reported to inhibit fat accumulation in 3T3-L1 adipocytes and suppressed the development of hyperglycemia via decreasing the SREBP-1c mRNA level in hepatocytes [65]. Another experimental study was conducted on pregnant rats (diabetes was induced by streptozotocin) in which the effects of reconstituted lyophilized powder and dehydrated mushrooms powder of Agaricus blazei, Ganoderma lucidum or Lentinula edodes at daily dose of 100mg/kg were investigated from gestation day 1 to 19. Mushrooms G. lucidum and L. edodes reduced glucose levels in glycemic curve in response to glucose tolerance test thereby suggesting potential of mushrooms as functional foods in management of diabetes [66].
In a clinical study, oral hypoglycaemic effect of freeze-dried suspensions and powder prepared from Pleurotus ostreatus and Pleurotus cystidiosus on both healthy and type 2 diabetic humans was investigated. A dose of 50mg/kg/body weight was administered to both groups while on a diet control, followed by a glucose load. A significant reduction (P < 0.05) in fasting and postprandial serum glucose levels of healthy volunteers and decreased postprandial serum glucose levels and increased the serum insulin levels (P < 0.05) of type 2 diabetic patients was observed. The researchers simultaneously conducted animal study to understand the mechanism of action using same dose. The medicinal mushrooms demonstrated possible increased glucokinase activity and insulin secretion that improved the utilization of glucose by peripheral tissues, inhibited glycogen synthase kinase and promoted glycogen synthesis [67].
A clinical study on 71 participants with type 2 diabetes evaluated the safety and efficacy of Ganopoly (polysaccharides extracted from G. lucidum) for 12 weeks at 1800mg thrice a day. Significant decrease in the mean glycosylated haemoglobin (HbA1c) was observed with decrease in post-prandial glucose. The study found Ganopoly to be efficacious and safe in lowering blood glucose concentrations [68]. A double blind, randomised, placebo-controlled trial evaluated the safety and efficacy of Ganoderma lucidum for the treatment of hyperglycemia and cardiovascular risk factors of metabolic syndrome. Ganoderma lucidum extract was used with or without Cordyceps sinensis in dose of 3 grams/day for 16 weeks. Eighty four, 18 years and older participants with type 2 diabetes mellitus and metabolic syndrome were randomised to one of three intervention groups – Ganoderma lucidum, Ganoderma lucidum with Cordyceps sinensis or placebo. The HbA1C and fasting plasma glucose were measured and the results were not found to be clinically significant or statistically significant on hyperglycemia, hypertension, or lipid profile in adults with metabolic syndrome or type 2 diabetes [16]. The effects on metabolic profile could be attributed to the presence of polysaccharides that are mainly classified as β-glucan polymers [owing to β-(1→3) linkages with some β-(1→6) branches along with presence of chitin, mannans, galactans, and xylans]. Pancreas enzymes are not equipped to hydrolyze the β-glucosidic bond, thereby allowing for prebiotics action of glucans that modulates gut microbiota, improves utilization, dietary energy, gene expression and gut permeability. Further polysaccharide glucan content of medicinal mushrooms [69],[70]:
-
Reduces serum glucose and lipoproteins;
-
Enhances serum insulin;
-
Improves liver PPAR-α expression and mRNA expression of protein levels;
-
Provides structural support to pancreatic β-cells;
-
Antioxidant and anti-inflammatory action.
Thus, medicinal mushrooms may have multiple metabolic actions that may cause improvement in insulin response and glucose homeostasis at cellular levels. It may also reduce underlying inflammation and strengthen epithelial and hepatic cells to prevent damage from any type of diabetes including GDM. However, there is insufficient evidence to draw definitive conclusions about the efficacy of individual medicinal mushrooms for Gestational diabetes unless human randomized trials are completed.
6.
Discussion
There is association between gestational diabetes and type 2 diabetes and it has been observed that women who have had gestational diabetes have at least seven-fold risk of developing type 2 diabetes in future compared to those women who have had a normal pregnancy. The risk factors are family history of diabetes, raised body-mass index, increased age, Asian ethnicity [3]. There are currently few clinical studies that have investigated the effects of mushrooms on diabetes or gestational diabetes mellitus in humans. The sample size of these studies was small, all dietary factors were not evaluated for confounding effects, various insulin and glucose control functions were not assessed consistently in all the studies which makes it difficult to formulate guidelines. Furthermore, there is not enough evidence on the dose, dosage form and side-effects of medicinal mushrooms that may be effective for pregnant females. According to American Herbal Products Association Botanical Safety Handbook, both mushrooms Ganoderma lucidum and Cordyceps sinensis are listed as safest drug class (Class 1 Drug) with no known herb-drug interactions. However, there is lack of scientific data on a safety and efficacy of Ganoderma dosage but 1.5 gms to 9 gms of dried extract per day is recommended by Chinese medicine experts [16]. There is immense scope to investigate the duration of supplementation to manage glucose irregularities in females with gestational diabetes. Studies are also required to investigate whether maternal supplementation of medicinal mushrooms influences growth and physiology of infants. There is also lack of consistency of clinical data for long term consumption to demonstrate the efficacy and safety in GDM females and also the quality control and standardisation data for the production of mushroom supplements is needed for its nutraceutical use [70].
7.
Conclusion
The current review highlights the potential of medicinal mushroom in management of Gestational diabetes: a disease with serious, long-term consequences for both baby as well as mother. The heterogeneity of the data from animal and human study prevented a statistical analysis/meta-analysis, hence a qualitative review was conducted. GDM is a growing health concern and evidence now suggests that screening, early detection and management can greatly improve outcomes for both baby and a mother. There is no uniformity in the screening and diagnostic methods around the world which leads to underdiagnoses and under management of gestational diabetes.
The present review has shown that polysaccharides, β-glucan, vanadium and other nutrients in mushrooms may address deranged metabolic profile in diabetics thereby improving immune responses, liver and pancreatic enzyme functions and exert prebiotic effect and improve gut permeability. Dietary source is always a popular and economical choice to manage a metabolic disorder in a community. Therefore, future research on mushroom species among humans is required that could assist in testing dosage and form of consumption for better health outcomes among gestational diabetic women.








 DownLoad:
DownLoad: