1. Introduction
The extracellular signal-regulated kinases ERK1/2 are key molecules regulating cell proliferation and gene expression, and are rapidly activated by a variety of hormonal and neural stimuli [1,2,3]. In pancreatic acinar cells, ERK1/2 are activated by a variety of secretagogues including cholecystokinin (CCK), acetylcholine (ACh), bombesin and Substance P [4,5,6,7,8,9]. The receptors for these secretagogues are all G protein coupled receptors and the CCK-A and muscarinic M3 receptors have been well studied [10,11]. These receptors couple with the heterotrimeric G proteins Gq/11 to activate phospholipase C β, and thereby produce inositol trisphosphate (IP3) and diacylglycerol (DAG); DAG will activate PKC [12].
ERK1/2 can be activated by a PKC-independent Ras-dependent pathway, or by a PKC-dependent Ras-independent pathway, depending on the receptor and cell type [1,13]. In dissociated pancreatic acinar cells and pancreas derived AR42J cells, phorbol esters activated ERK1/2 similar to CCK, and the general PKC inhibitor GF109203X inhibited both TPA-and CCK-induced ERK1/2 activation; down-regulation of PKC by long term treatmentwith phorbol ester also inhibited CCK-induced ERK1/2 activation [5,6,7]. Furthermore, overexpression of dominant-negativeRas did not inhibit CCK-induced ERK1/2 activation in rat acini [14]. Therefore, CCK-induced activation of ERK1/2 appears to be primarily PKC-dependent and Ras-independent in pancreatic acinar cells. These data suggest that PKC plays an important role in ERK1/2 activation by secretagogues in pancreatic acinar cells. By contrast, epidermal growth factor (EGF) activates ERK1/2 through PKC-independent mechanism most likely involving Ras [5].
Protein kinase C (PKC) is a group of phospholipid-dependent, serine/threonine kinases implicated in various biological functions including proliferation, differentiation and secretion [15,16]. The PKC family is divided into three subgroups based on their structure and activation requirements: conventional PKCs (α, βⅠ, βⅡ and γ) which are DAG and calcium-dependent; novel PKCs (δ, ε, η and θ), which are DAG-dependent but calcium-independent; and atypical PKCs (ζ, λ/ι and μ), which are not activated by DAG [15,16]. In the four constant (C) domains of PKC, the C1 domain contains the binding site for DAG or phorbol ester, the C2 domain contains the calcium-binding site, the C3 domain contains the ATP-binding site and the C4 domain contains the protein substrate recognition site. In addition, many isoforms of PKC are further regulated by phosphorylation and by localization within cells [16,17]. It is well accepted that translocation to a membrane fraction is tightly associated with activation of PKCs. PKCs translocate to specific subcellular sites after activation through direct interaction with docking proteins named Receptors for Activated C Kinases (RACKs) [17], which allow specific substrate recognition and signal transduction. Different PKC isoforms have their own RACKs and peptides derived from the RACK sequence which block the interaction of a PKC with its RACK can act as PKC inhibitors [18,19,20].
Different cell types express their own unique subset of PKC isoforms and the expression of multiple isoforms regulating diverse functions within a single cell type suggest that individual PKC isoforms may have specific functions. Four PKC isoforms have been identified in rodent pancreatic acinar cells: α, δ, ε, and ζ
[21,22,23,24,25] while human acinar cells posess additional isoforms [25]. Different PKC isoforms have been linked to specific functions and signal transduction pathways in pancreatic cells. While earlier work used chemical inhibitors, more recent studies have used cell permeant peptides that disrupt binding to RACKs and dominant negative kinases delivered by adenoviruses. In the present study, using chemical inhibitors, cell permeant inhibitory peptides and adenoviral delivered PKC molecules, we compared the role of different PKC isoforms in ERK1/2 activation; our results indicate that PKC ε mediates ERK1/2 activation in rat pancreatic acini.
2. Materials and Methods
2.1. Materials
Sulfated CCK octapeptide was obtained from Research Plus (Bayonne, NJ); carbamylcholine chloride (carbachol), bombesin, phorbol 12-myristate 13-acetate(PMA) and soybean trypsin inhibitor (SBTI) were from Sigma Chemical (St. Louis, MO). Acinar isolation used either chromatographically purified collagenase (CLSPA from Worthington Biochemicals, Freehold, NJ) or collagenase from Crescent Chemical (Islandia, NY). The PKC inhibitors GF109203X and Go6976 were from Calbiochem (La Jolla, CA) while Rottlerin was obtained from Biomol Research Laboratories (Plymouth Meeting, PA). Polyclonal antibodies against PKC α, δ, ε and ζ were from Research & Diagnostic Antibodies (Benicia, CA), Polyclonal antibody against phospho-ERK1/2 was from Cell Signaling Technology (Beverly, MA), Monoclonal antibody against β-actin was from Sigma (St. Louis, MO) and Raf antibodies were from Santa Cruz Biotech (Santa Cruz, CA). Peptide regulators of PKC isoforms coupled by disulfide bonds to a cell permeant Antennapedia peptide were obtained from Dr. D. Mochly-Rosen (Stanford University, Palo Alto, CA) and have been described previously [18,19,20]. Adenoviruses encoding wild type and dominant negative PKC α, δ and ε were a gift from Dr. J. Molkentin (University of Cincinnati, Cincinnati, OH) and have been described previously [26].
2.2. Preparation of isolated pancreatic acini, short-term culture, and viral infection of pancreatic acini
Pancreatic acini were isolated by collagenase digestion of pancreas from 125 to 150 g male Sprague-Dawley rats as previously described [23,27]. Acini were suspended at 37 ℃ in HEPES-Ringer (HR) buffer supplemented with 11.1 mM glucose, Eagle's minimal essential amino acids, 0.1 mg/mL SBTI, 1 mg/mL BSA, equilibrated with 100% O2 andstudied within 2 h using chemical inhibitors and 6 h for inhibitory peptides. Acini were treated with chemical inhibitors (5 μM) 30 min before stimulation while inhibitory peptides (1 μM) were added to HR buffer 6 h and again at 30 min before stimulation. In the viralinfection experiments, isolatedacini were resuspended in Dulbecco's modifiedEagle's medium (DMEM) enrichedwith 0.5% fetal bovine serum and antibiotics, placed in 100 mm petri disheseach containing 10 mL media, and incubated overnight for 14–16 h at 37 ℃ in a 5% CO2 environment. Adenoviruses prepared and tittered as described earlier [23,27] and added at a specified titer, usually 5 × 106 pfu/mL, to the culture medium at the beginning of the overnightincubation. Overnight-cultured acini were washed and then resuspended in HR buffer for 30 min before stimulation.
2.3. Western blotting and immunoprecipitation
Lysates of cultured acini were prepared in buffer containing 0.5% Triton X-100 and Western blotting was carriedout as previously described [23,27] applying 50 μg of lysate protein to each lane. To confirm equal loading, membranes were striped and reprobed with anti β-actin antibody. ECL chemiluminescence reagentswere used to visualize the secondaryantibody with attention paid to ensure film was not saturated. Films were scanned and band density quantitated using Multi-Analyst software (Bio-Rad). For immunoprecipitation studies, aciniwere pelleted after treatment, washed once with 1 mL ice-cold PBS containing1 mM Na3VO4 (pH 7.4) and then resuspended in 500 μL ice-coldlysis buffer (50 mM Tris, 150 mM NaCl, 5 mM EDTA, 25 mM sodiumfluoride, 1% Triton X-100, 1 mM PMSF, 0.2 mM Na3VO4, 1 mM dithiothreitol, 10 μg/mL leupeptin, 10 μg/mL aprotinin pH 7.4) and sonicated.After cell lysis, samples were centrifuged at 12,000 g for 15min at 4 ℃ and the protein concentrations in the supernatants were measured and adjusted to 1 mg/mL. One microgramof anti-c-Raf-1 was added to each sample with 1 mg acinar protein and the mixture was incubated for 2h at 4 ℃. The immunocomplexes formed were precipitatedby incubation with 30 μL of protein-A agarose beads at 4 ℃ for 1 h. Finally, immunoprecipitates were washedthree times with iced lysis buffer, solubilized and subjected to SDS-PAGE and Western blotting with specific antibodies.
2.4. Statistical analysis
The data presented are the mean ± S.E. of 3 to 6 separate experiments. Statistical significance was calculated by the Student's t test with p < 0.05 representing significance.
3. Results
3.1. Overnight treatment with PMA down-regulated PKC α, δ, and ε and inhibited CCK-induced ERK1/2 activation
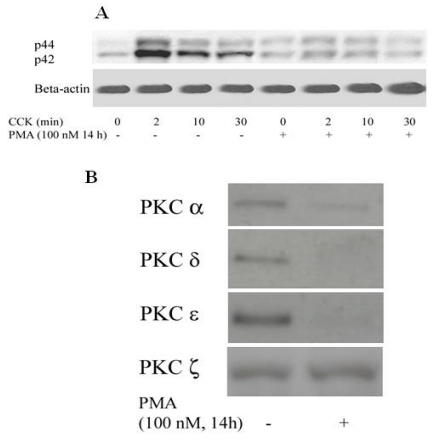
To determine whether PKC participated in CCK-induced ERK1/2 activation in rat pancreatic acini and which isoform could mediate this effect, we first down-regulated PKC protein with PMA. In overnight-cultured acini, ERK1/2 activation (phosphorylation) stimulated by CCK peaked at 2 min and then declined, but stayed, at higher than control level out to 90 min (data not shown). PMA 14 h treatment had no significant effect on basal ERK1/2 phosphorylation, but CCK-induced ERK1/2 activation was significantly inhibited (Figure 1A). Our previous data indicated that there were four PKC isoforms (α, δ, ε and ζ) detected in rat pancreatic acinar cells and three of them (α, δ and ε) were activated by CCK [23]. Overnight treatment with 100 pM PMA significantly reduced the protein level of PKC α, δ and ε, but not the ζ isoform (Figure 1B). Since PMA treatment blocked ERK1/2 activation but did not down regulate PKC ζ, and because CCK did not activate PKC ζ in acinar cells [23], PKC ζ does not appear to be involved in CCK-induced ERK1/2 activation.
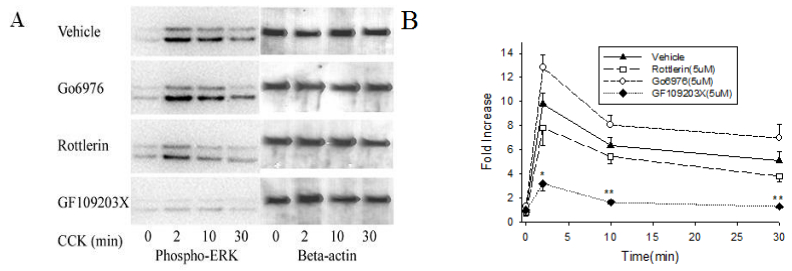
3.2. Effect of three chemical inhibitors on CCK-induced ERK1/2 activation
The broad-spectrum PKC inhibitor GF109203X, which blocks conventional and novel PKCs and therefore should inhibit α, δ and ε, inhibited ERK1/2 activation at a concentration of 5 μM (Figure 2). At the peak activation time of 2 min, GF109203X decreased the increase in ERK1/2 activity from 9.8 ± 0.9-fold to 3.2 ± 0.6-fold. Basal activity was subtracted from stimulated values and subsequently the stimulated value in the presence of GF109203X compared to those with GF109203X absent; the decrease with GF109203X was 75% at 2 min, 88% at 10 min and 92% at 30 min. The conventional PKC inhibitor Go6976, which should inhibit α but not δ and ε, showed no inhibitory effect at 5 μM (Figure 2). Interestingly Go6976 actually enhanced ERK1/2 activation, but this effect was not significant. The PKC δ inhibitor rottlerin showed no significant inhibitory effect at any time (Figure 2). The sample was re-blotted with anti-β-actin or in some experiments with anti-total ERK1/2 to serve as a control for equal loading. All three PKC inhibitors showed no significant effect on basal ERK1/2 activity.
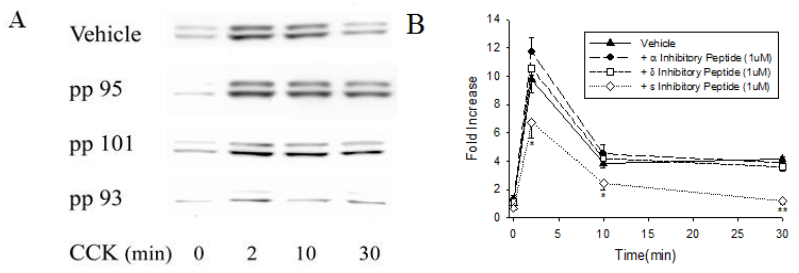
3.3. Isoform-specific PKC ε inhibitory peptide inhibited CCK-induced ERK1/2 activation
Inhibitory peptides for PKC α,
δ, ε, and vehicle were added to HR buffer for 6 to 7 h before CCK stimulation. The PKC ε inhibitory peptide inhibited the CCK-induced ERK1/2 activation by 33 % from 9.7 ± 0.9-fold to 6.7 ± 1.1-fold at 2 min; at 10 and 30 min, the ERK1/2 response was decreased by 45 and 91% respectively (Figure 3). The other peptides designed to inhibit PKC α and δ and the scrambled control peptide did not affect CCK-induced ERK1/2 activation at any time (data not shown). The effect of PKC ε inhibitory peptide was smaller then that of GF109203X at 2 min, but similar at 30 min. We did not use more than 1 μM because more Attennapedia peptide could have a deleterious effect. All inhibitory peptides had no significant effect on basal CCK activation level even though PKC ε inhibitory peptide decreased basal by 13% (Figure 3). This data indicates the possible role of PKC ε in ERK1/2 activation.
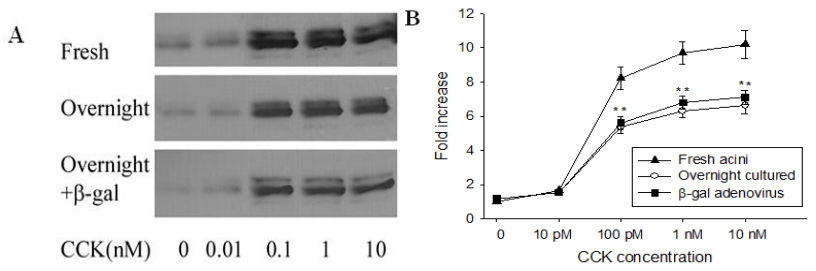
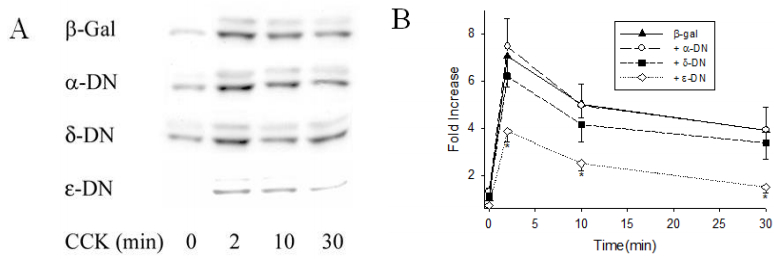
3.4. Dominant negative PKC ε mutant inhibited CCK-induced ERK1/2 activation
There are known to be some differences between fresh acini and overnight-cultured acini [27]. Our earlier study demonstrated that there was a reduced amylase release as percent of total and a 3-fold increase in the concentration of CCK required for maximal amylase release in overnight-cultured rat pancreatic acini [23]. For ERK1/2 activation, we also noticed a reduced activation level in cultured rat pancreatic acini, although no change in the concentration of CCK was required (Figure 4). The 100 pM CCK-induced ERK1/2 activation of overnight-cultured rat acini decreased to 6.3 ± 0.4-fold from 9.7 ± 0.7-fold for fresh rat acini. Infection of rat acini by an adenoviral vector expressing β-gal had not effect beyond that of overnight culture (Figure 4).
The dominant negative PKC α, δ and ε cDNAs contain a lysine to arginine mutation in the ATP binding domain at amino acid positions 368,376 and 436 respectively. In our earlier work, we showed that when the incubation time was fixed at 14 h and with 5 × 106 pfu/mL titer, all different PKC dominant negative expression were 5 to 10-fold higher than in acini infected with b-gal as a control; and that expression of a dominant negative PKC showed no effect on expression of other PKC isoforms [23]. In the present study, overexpression of dominant negative PKC ε inhibited CCK-induced ERK1/2 activation by 50% from 7.0 ± 0.7 to 3.9 ± 0.5-fold at 2 min; there was also a 56% decrease at 10 min and 73% decrease at 30 min (Figure 5). Overexpression of dominant negative PKC α and PKC δ had no significant effect on CCK-induced ERK1/2 activation at any time. Overexpression of dominant negative forms of PKC α and δ had no significant effect on basal ERK1/2 activity; PKC ε dominant negative lowered the basal level by 29%, but this was not statistically significant (Figure 5).
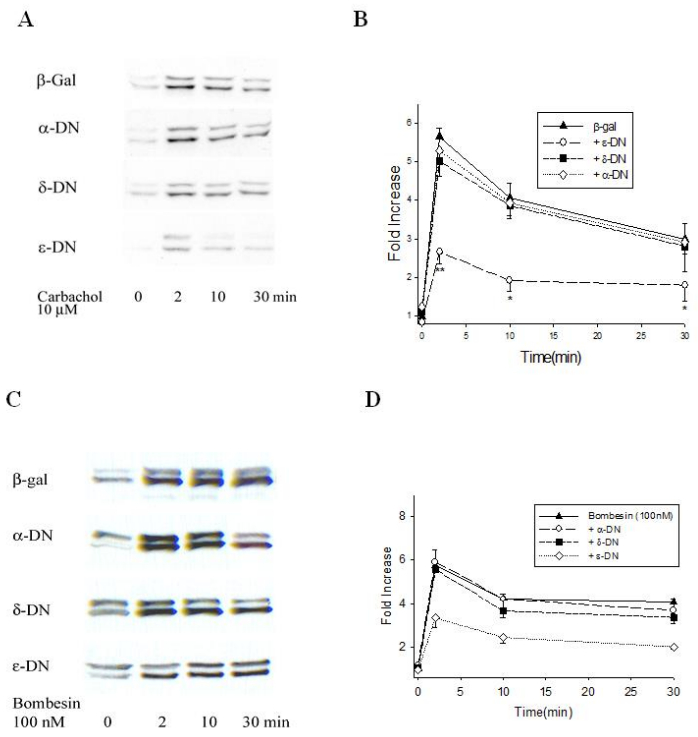
To determine whether the dominant negative inhibition was receptor specific, we also studied the effect of dominant negative PKC μutants on carbachol-and bombesin-induced ERK1/2 activation and the results were very similar to CCK as only PKC ε dominant negative inhibited ERK1/2 activation (Figure 6). For carbachol stimulation at 2, 10 and 30 min, there were 61, 68 and 52% deceases respectively. For bombesin stimulation, ERK1/2 activation was decreased at 2, 10, 30 min by 50, 54 and 66% respectively. In these experiments PKC ε dominant negative also lowered the basal level, but this was not statistically different.
3.5. PKC ε mediated ERK1/2 activation through the interaction with c-Raf-1
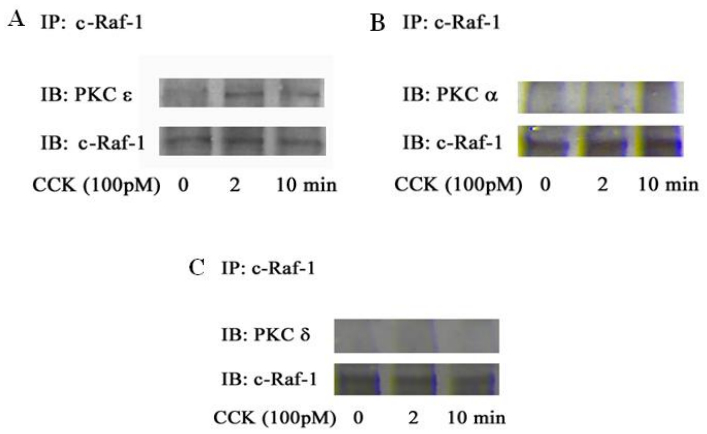
Using immunoprecipitation and Western blotting, we found that PKC ε interacted with c-Raf-1 upon CCK stimulation, (Figure 7A). We also found that c-Raf-1 did not interact with PKC α and δ after CCK stimulation (Figure 7B and C). CCK is known to activate all three Raf forms: Raf-A, Raf-B and c-Raf-1 [5]. In other experiments Raf-A and Raf-B were immunoprecipitated and there was no association with PKC ε (data not shown). Thus, the most likely pathway for ERK1/2 activation involves PKC ε activation of c-Raf-1.
4. Discussion
CCK is known to activate multiple isoforms of PKC in pancreatic acinar cells [22,23,24,25]. Because multiple isoforms are present in most cells it seems likely that they exist to control different functions. The present study focused on discriminating the PKC isoform involved in activating ERK in response to CCK. Using both isoform specific peptide inhibitors and overexpression of dominant negative PKC isoforms we show that PKC-ε is the primary isoform mediating ERK activation in rat acinar cells. The determination in acinar cells is important since in other cell types different PKC isoforms have been related to ERK activation although most studies show mediation by one of the Ca2+-independent isoforms especially δ and ε [26,28,29,30,31,32].
Our initial studies used down regulation of PKC by PMA which eliminated PKCζ as a potential activator as it was not down regulated and down regulation blocked ERK activation. We then evaluated chemical inhibitors. The inhibitory effects of all the chemical PKC inhibitors are due to an interaction with the ATP-binding site of PKC [33,34]. At μM concentrations, GF109203X is known to totally block PKC α, δ and ε activity, but has almost no effect on PKC ζ [34]. GF109203X almost totally blocked CCK-induced ERK1/2 activation. These results confirmed earlier studies that CCK-induced ERK1/2 activation is PKC-dependent [4,5,6]. Go6976, which inhibits PKC α at nM concentrations had no significant inhibitory effect on ERK1/2 activation at 5 μM, but rather some enhancement. However, other means of inhibiting of PKC α did not show this enhancement effect. It is therefore likely that PKC α does not mediate activation of ERKs in acini. The PKC δ inhibitor rottlerin also did not show significant inhibitory effect at 5 μM. Subsequent data, however, has questioned the specificity of rottlerin on pancreatic acini [35]. Thus, the use of rottlerin by itself can not allow a definitive determination.
Because of the uncertain specificity of chemical inhibitors and because there is no PKC ε inhibitor available at present, we utilized isoform specific peptide inhibitors to identify the PKC isoform mediating ERK1/2 activation. All the synthesized peptide inhibitors are short peptides that include the isoform specific RACK binding site and have been shown to block the interaction between PKC and their RACK [20,36,37,38,39]. The peptides have a Cys residue at their amino termini by which they are cross-linked by a Cys-Cys bond to Drosophila Antennapedia homeodomain-derived carrier peptide which is a cell permeant carrier peptide [20]. Only the PKC e inhibitory peptide had an inhibitory effect on CCK-induced ERK1/2 activation suggesting that PKC e is the major isoform mediating ERK1/2 activation.
To confirm the peptide inhibitor results by a different approach, we also utilized an adenoviral delivery system to express dominant negative PKC α, δ and ε to clarify the role of specific PKC isoforms. The dominant negative PKC mutants consist of a lysine to arginine substitution in ATP binding domain. Although they are inactive, they still can interact with endogenous RACKs, thereby blocking the action of endogenous PKC isoforms to interact with the same RACKs [26]. Adenoviral infection has been shown to be a highly efficient method of gene transfer to pancreatic acinar cells with nearly 100% efficiency [14,23,27]. In this study, 14-16 h incubation was used to obtain adequate expression of PKC protein, and also to preserve as much as possible the acinar polarity and secretory responsiveness. Various adenoviral titers were tested and 5 × 106 pfu/mL was used as standard. At this titer, there is a 5 to 10-fold increase of protein expression compared to control without affecting the expression of other PKC isoforms, each PKC dominant negative inhibited endogenous activity of that isoform specifically [23]. When fresh and overnight-cultured pancreatic acinar cells were compared, there was decreased ERK1/2 activation level at same CCK concentration, but no maximal concentration change required for ERK1/2 activation (Figure 3). Only PKC ε dominant negative expression inhibited the ERK1/2 activation. To show that this effect was not specific to the CCK receptor, we used carbachol and bombesin which act on distinct receptors to activate PKC. The PKC ε dominant negative also inhibited carbachol-and bombesin-stimulated ERK1/2 activation indicating that the effect of PKC ε dominant negative is not specific for CCK receptor. That the PKC δ dominant negative is in fact functional is shown by the fact that it can selectively inhibit amylase release [23]. In experiments on mouse acinar cells in monolayer culture, dominant negative PKC α, as well as PKC ε, inhibited DNA synthesis, indicating that PKC α dominant negative is also active (L. Guo and J.A. Williams, unpublished data).
It has been reported in pancreatic acinar cells that CCK-induced ERK1/2 activation is Ras-independent [5,14] and CCK can activate all three Raf family members [5]. A study in the pancreas derived cell line AR42J showed that PKC ε interacted with c-Raf-1 upon CCK stimulation [8]. In this cell thype, however, CCK activates a CCK2 (Gastrin) receptor which is known to activate Ras. Therefore, it is possible that PKC ε mediates ERK1/2 activation via interaction with c-Raf-1 in differentiated pancreatic acinar cells. In rat acini, our coimmunoprecipitation data showed that PKC ε interacted with c-Raf-1 after CCK stimulation, but not Raf-A or Raf-B. c-Raf-1 did not interact with PKC α and δ upon CCK stimulation (Figure 7). Raf-B has been shown to mediate the effects of cyclic AMP via Rap1 to activate ERK in some cell types but this pathway does not appear to activate ERK in acinar cells [40]. The mechanism by which PKC activates Raf is also not fully understood. PKC phosphorylates cRaf-1 at multiple sites but these sites do not appear to be necessary. One mechanism is phosphorylation of RKIP (Raf kinase inhibitory protein) on Ser153 which causes dissociation with resulting activation of Raf [41].
In conclusion, the current study demonstrates that PKC ε plays an important role in regulating pancreatic ERK1/2 activation induced by CCK most likely through interaction with c-Raf-1. In pancreatic acini, it seems that each PKC isoform has its unique function. While PKC ε appears involved in CCK-induced ERK1/2 activation, PKC δ and -ε mediate NF-κB activation [42,43] and participate in the premature activation of trypsinogen in overstimulation pancreatitis [44]. PKC-δ has also been related to activation of PKD1. PKC is well established as a mediator of physiological amylase secretion. One study indicated that PKCdelta was the specific isoform involved in amylase secretion [23] as has been suggested for pancreatic beta cells [45]. However, a study of PKC delta gene deleted mice failed to confirm this [46]. Thus, the isoform participating in amylase secretion is unclear at present. While PKC a is involved in proliferation in other cell types its physiological function in acinar cells is not yet established although following supraphysiological stimiulation it mediates phosphorylation of Munc-18c and basolateral exocytosis [47]. While not yet fully elucidated the multiplicity of PKC isoforms in acinar cells appears to be related to different roles in the regulation of diverse physiological effects.
Acknowledgements
This research was supported by NIH grants DK41122 and DK52860 to JAW and by Michigan Gastrointestinal Peptide Center, P30-DK34933. We thank Margaret Westfall and Jeff Molkentin for the gift of the adenoviral vectors.
Conflict of interest
All authors declare no conflicts of interest in this paper.








 DownLoad:
DownLoad: