1. Introduction
With rising incidences associated with an epidemic in obesity and metabolic syndrome, pancreatic cancer is expected to become the second most common cause of cancer-related deaths by 2030 [1]. It is characterized by rapid disease progression without peculiar symptoms and the five-year overall survival (OS) is very limited [2]. Surgery offers the best chances of survival for the patients; however, it is possible only in a small percentage of cases, since diagnosis of pancreatic cancer often occurs late. In addition, even resectable pancreatic cancers display a high rate of recurrence [3]. Systemic chemotherapy includes the nucleoside analog gemcitabine (Gem), that is the only approved single-agent chemotherapeutic drug for locally advanced and metastatic pancreatic cancer [4]. However, the response to chemotherapy is far from being satisfying. Moreover, approaches other than systemic chemotherapy, such as radiotherapy, targeted therapies and non-conventional therapies have also failed to produce valuable clinical benefits up to now [2,3,4]. Thus, novel therapeutic options to fight this deadly cancer are urgently warranted. Combination chemotherapy is receiving close attention in order to find compounds that increase the therapeutic index of clinical anticancer drugs [5,6]. Naturally occurring molecules with antitumor activity and with the least toxicity to normal tissues are proposed as intriguing candidates to be investigated for their synergistic efficacy in combination with conventional antineoplastic drugs [5,6,7,8].
Among the natural compounds from medicinal plants, forskolin can be suggested as one of the most interesting molecules to possibly use in cancer therapy [9]. Forskolin is a diterpene produced by the roots of the Indian plant Coleus forskohlii [10]. Notably, the natural compound forskolin has been used for centuries in traditional medicine and its safety has also been documented in conventional modern medicine [11,12,13]. Forskolin directly activates the adenylate cyclase enzyme, that generates cAMP from ATP, thus raising intracellular cAMP concentrations. It is largely known that cAMP, either via PKA-dependent or PKA-independent mechanisms, affects numerous cellular functions and is considered very relevant to cancer [14,15]. Importantly, the cAMP elevating agent forskolin has been shown to cause numerous relevant anticancer effects, such as the induction of mesenchymal-to-epithelial transition, the enhancement of the sensitivity to conventional antineoplastic drugs and the inhibition of proliferation, motility and migration in many types of cancer cells, including pancreatic cancer ones [16,17,18,19,20,21,22,23].
Here we investigate the potential anti-pancreatic cancer activity by forskolin and, particularly, evaluate whether forskolin can increase the chemosensitivity to gemcitabine of pancreatic cancer cells and the underlying signaling mechanisms.
2. Materials and Methods
2.1. Drugs and reagents
Dulbecco's modified Eagle's medium (DMEM), propidium iodide (PI) and 3-(4, 5-dimethylthiazol-2-yl)-2, 5-diphenyltetrazolium bromide (MTT) were purchased from Sigma Life Science (Milan, Italy). Phosphate-buffered saline (PBS) and trypsin-EDTA were purchased from Lonza Verviers Sprl, (Belgium). Bovine serum albumin (BSA), and fetal bovine serum (FBS) were obtained from Gibco (Grand Island, NY). Tissue culture dishes were purchased from Microtech (Naples, Italy). Annexin V-fluorescein isothiocyanate (V-FITC) Apoptosis Detection kit was purchased from eBioscience (San Diego, CA, USA). Anti-tubulin antibodies were obtained from Oncogene-Calbiochem (La Jolla, CA). Anti-p-ERK, anti-FAK, anti-p-FAK antibodies were obtained from Cell Signaling Technology (Danvers, MA). All other antibodies were obtained from Santa Cruz Biotechnology (San Diego, CA). Blots were incubated with horseradish peroxidase (HRP) conjugated goat anti-rabbit (Immunoreagents Inc., GtxRb-003-DHRPX) secondary antibodies. Forskolin and Gemcitabine were purchased from Sigma (Sigma-Aldrich, St. Louis, MO). All buffers and solutions were prepared with ultra-high quality water. All reagents were of the purest commercial grade.
2.2. Cell culture and treatments
Panc-1 and AsPC-1 human pancreatic cancer cell lines were obtained from the American Type Culture Collection (Rockville, MD) and as a generous gift from Dr Alfredo Budillon, respectively. Panc-1 and AsPC-1 cells were grown in Dulbecco's Modified Eagle's Medium (DMEM) supplemented with 2mM glutamine, 100 U/mL penicillin, 100 mg/mL streptomycin and 10% fetal bovine serum (FBS) and cultured at 37 ℃ in a 5% CO2 humidified atmosphere. Gemcitabine was dissolved in ddH2O, stored at 4 ℃ and diluted with culture medium to final concentrations indicated in the figures. Forskolin was prepared as stock solution in DMSO and dilutions were made such that final concentration of solvent was kept below 0.1%. Control cells were treated with an equivalent volume of solvents. Typically, cells were split (6 × 105/10 cm plate) and grown in 10% serum containing medium. After 24 h, the medium was removed, the cells were washed with PBS and incubated with 10% FBS fresh medium (Time 0), supplemented or not with forskolin and gemcitabine, alone or in combination, and grown for the times and at concentrations indicated in the text and figures. After treatment, both floating cells and adherent cells were recovered and counted (by centrifugation from culture medium and harvested by trypsinization, respectively). Both floating and adherent cells were used in the experiments aimed to perform fluorescence-activated cell sorting (FACS) analysis.
2.3. In vitro wound-healing cell migration assay
Cell migration assays were performed as described previously [24]. Typically, cells were split (5 × 105/6 cm plate) and grown in 10% serum containing medium. After 24 hours, the medium was removed, cells were washed with PBS and cultured in 10% serum fresh medium. Approximately 36–48 h later, when cells were 95–100% confluent, the medium was removed, cells were washed with PBS and cultured in low-serum (0.1%) fresh medium. After 24 hours of serum starvation, a gap was introduced into the monolayer cells by dragging a yellow tip across the cell surface. Medium and not adherent cells were removed, and cells were washed twice with PBS, and new 0.1 or 10% serum containing medium without (control) or with forskolin was added. Cells were permitted to migrate into the area of clearing for up to 48 hours. Wound closure was monitored by visual examination under microscope. The phase contrast images of the wounds were recorded after different times for up to 48 hours at the identical location of the initial image, and the wound closure was evaluated by measuring the width of the remaining wound. All experiments were performed at least three times.
2.4. Cell viability assay
Cell viability assay were performed as described previously [25]. Briefly, cells were seeded in 10% FBS-containing medium in a 96-well plate at the density of 3 × 103 cells/well for 24 h at 37 ℃ and then cultured as described in "Cell culture and treatments". Cell viability was assessed by adding MTT solution in PBS to a final concentration of 0.5 mg/mL. The plates were then incubated at 37 ℃ for 3 h and the MTT-formazan crystals were solubilized in a solution of 4% 1N isopropanol/ hydrochloric acid at 37 ℃ on a shaking table for 20 min. The absorbance values of the solution in each well were measured at 570 nm using a Bio-Rad 550 microplate reader (Bio-Rad Laboratories, Milan, Italy). Cell viability was expressed as a percentage of absorbance values in treated samples respect to that of control (100%). MTT experiments were performed in quadruplicate.
2.5. Evaluation of sub-G1 and cell cycle phases by flow cytometry
After treatments, the cells were recovered as previously described in "Cell culture and treatments", fixed by resuspension in 70% ice-cold methanol/PBS and incubated overnight at 4 ℃. The cells were then centrifuged at 400× g for 5 min, and pellets obtained were washed with ice-cold PBS, centrifuged for 5 min, resuspended in 0.5 mL DNA staining solution (50 μg/mL PI and 100 μg RNase A in PBS), and incubated overnight at 37 ℃ in the dark. Samples were then transferred to 5-mL Falcon tubes and stored in ice until assayed. Flow cytometry analysis was performed using a FACSCalibur flow cytometer (Becton Dickinson, San Jose, CA, USA) interfaced with a Hewlett-Packard computer (model 310) for data analysis. For the evaluation of intracellular DNA content, at least 20,000 events for each point were analyzed, and regions were set up to acquire quantitative data of cells that fell into the normal G1, S, and G2/M regions and compared also with the events with fragmented DNA (sub-G1 or apoptotic events). Results were analyzed using ModiFIT Cell Cycle Analysis software.
2.6. Flow cytometry analysis of cell death
Cell death were measured by Propidium Iodide uptake and flow cytometry as previously described [26].
2.7. Preparation of cell lysates
Briefly, 3–5 volumes of RIPA buffer (PBS, 1% NP-40, 0.5% sodium deoxycholate, 0.1% SDS) containing 10 μg/mL aprotinin, leupeptin, and 1 mM phenylmethylsulfonyl fluoride (PMSF) were added to recovered cells. After incubation on ice for 1 hour, samples were centrifuged at 18.000 g in an Eppendorf microcentrifuge for 15 min at 4 ℃ and the supernatant (SDS total extract) was recovered. Some aliquots were taken for protein quantification according to Bradford method; others were diluted in 4× Laemmli buffer, boiled and stored as samples for immunoblotting analysis.
2.8. Immunodetection of proteins
Typically, we employed 20–40 μg of total extracts for immunoblotting. Proteins from cell preparations were separated by SDS-PAGE and transferred onto nitrocellulose sheets (Schleicher & Schuell, Dassel, Germany) by a Mini Trans-Blot apparatus BioRad (Hercules, CA). Ⅱ goat anti-rabbit or anti-mouse antibodies, conjugated with horseradish peroxidase (BioRad), were used as a detection system (ECL) according to the manufacturer's instructions Amersham Biosciences (UK).
2.9. Statistical analysis
Most of experiments were performed at least three times with replicate samples, except where otherwise indicated. Data are plotted as mean ± SD (standard deviation). The means were compared using analysis of variance (ANOVA) plus Bonferroni's t-test. P values of less than 0.05 were considered significant. National Institutes of Health Image J 1.42Q (NIH, Bethesda, MD) software were used for densitometric analysis.
3. Results
3.1. Forskolin inhibits migration of human pancreatic cancer cells
The human pancreatic cancer cell lines PANC-1 and AsPC-1 are well-established and widely used model systems of pancreatic cancer cells [22,23,27,28,29,30]. Throughout our in vitro experiments, we have used a spectrum of final concentration of forskolin up to 10 µM (not toxic, submaximal dose), in agreement with most of the studies regarding in vitro effects by forskolin, ranging from 1 to 100 µM [16,17,18,19,20,21,22,23].
Recently, it has been shown that in Panc-1 cells, 22-h treatment with 10 µM forskolin in serum-free medium was sufficient to arrest cellular migration compared to cells without treatment [31]. In the same study, authors also show that proliferation and apoptosis were unaffected by 10 µM forskolin in serum-free medium treatment up to 72 hours.
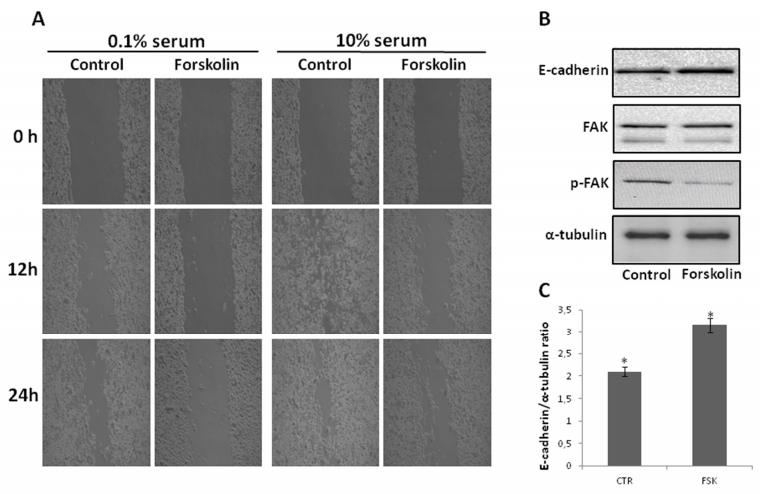
In order to further evaluate and extend the effects of cAMP elevating agent forskolin on migration of pancreatic cancer cells, we firstly looked at its impact on the migration of Panc-1 cells normally growing in 10% serum-containing medium. To this purpose, Panc-1 cells were treated or not with 10 µM forskolin for up to 24 hours both in 0.1 and 10% serum-containing medium and the migration rate of the cells was mesaured by wound-healing assay (Figure 1). In the qualitative in vitro wound-healing assay (Figure 1A), cells were grown to confluence. A wound was made in the middle of the culture plate with a pipette tip. As expected [31], control 0.1% serum Panc-1 cells covered (not completely) the wound after 24 hours (and even late); notably, high serum concentration increased cell migration, as in the culture plate cultured with 10% serum, a larger part of the wound was covered by cells after 24 hours (and even before). According to previous data regarding the anti-migratory effects of forskolin in Panc-1 cells [22,23,31], 10 μM forskolin impressively inhibited both basal and, more importantly, serum-induced cell migration. Similar results were obtained also with AsPC-1 cells (data not shown). To increase the reliabiity of the above data, Panc-1 cells treated or not with forskolin in 10% serum containing medium for 24 hours, were collected by scraping, total cell extracts in SDS-containing buffer were prepared, and protein levels of e-cadherin and of Focal Adhesion Kinase, FAK (along with its phosphorylation/activation status) were monitored by western blotting analysis. Consistent with the largely known relevant pro-migration role of FAK and anti-migration role of e-cadherin [32,33], very interestingly, we found that e-cadherin was increased and FAK phosphorylation level strongly decreased, upon forskolin treatment, respectively (Figure 1B).
Overall, the above data indicate that forskolin has inhibitory effects on basal and serum stimulated pancreatic cancer cell migration.
3.2. Forskolin inhibits proliferation of human pancreatic cancer cells
In order to further evaluate the effects of cAMP elevating agent forskolin on behaviour of pancreatic cancer cells, we looked at its impact on the proliferation of Panc-1 and AsPC-1 cells.
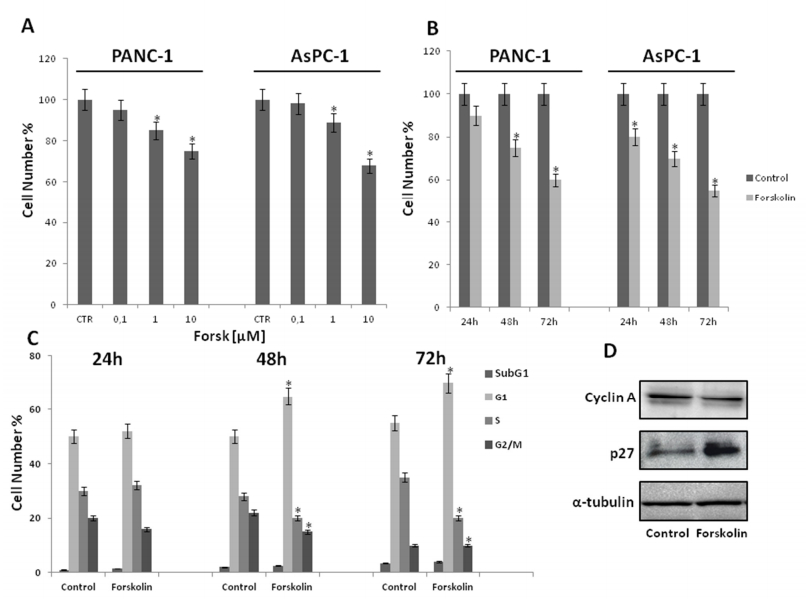
To this purpose, we performed dose-response and time-course experiments. Panc-1 and AsPC-1 cells were incubated in 10% serum containing medium with or without increasing concentrations (0.1, 1, 10 µM) of forskolin for 48 hours and then cell proliferation was investigated by both direct cell number counting as well as by conventional MTT assay. As shown in Figure 2A, forskolin causes a dose-dependent reduction of the number of Panc-1 and AsPC-1 cells (statistically significant, p < 0.05) starting from the dose of 1 µM. Similar results were obtained by MTT assays (data not shown).
Next, we performed time-course experiments. Panc-1 and AsPC-1 cells were exposed or not to forskolin 10 µM in 10% serum containing medium for 24, 48 and 72 hours, after which cell proliferation was determined by conventional MTT assay and by direct cell number counting. As reported in Figure 2B, forskolin causes a time-dependent reduction of cell number (p < 0.05) already evident at 24 hours and very clear at 48 and 72 hours, respectively (Figure 2B). According results were obtained by MTT assays (data not shown).
To further explore the growth inhibitory effect of forskolin on pancreatic cancer cells, distribution of Panc-1 and AsPC-1 cells in the cell cycle phases was evaluated by flow cytometric analysis of propidium iodide-stained cells in response to forskolin 10 µM treatment. We also looked at the proportion of cells with hypodiploid DNA content (sub-G1 population), characteristic of cells having undergone DNA fragmentation which is a biochemical hallmark of apoptotic cell death. Figure 2C shows that time-dependently (from 24 to 72 hours), in forskolin-treated cells the percentage of G1 phase cells is higher than that of control untreated cells, with a concomitant decrease of S and G2/M phases. Moreover, in Figure 2C it is also shown that no appearance of the sub-G1 population in response to forskolin treatment was observed up to 72 h. In addition, we also looked at the cell cycle inhibitor p27kip1 and cyclin A protein levels; as expected, in agreement with the G1 phase accumulation in response to forskolin, p27 cell cycle inhibitor and cyclin A protein levels increase and decrease, respectively (Figure 2D). Similar results were obtained also with AsPC-1 cells (data not shown).
In agreement with previous results [34], altogether these data indicate that forskolin has inhibitory effects on pancreatic cancer cell proliferation, mainly by slowing down the cell division cycle and not inducing cell death.
3.3. Forskolin sensitizes pancreatic cancer cells to gemcitabine
In 1997, a randomized phase Ⅲ study of gemcitabine versus 5-fluorouracil provided evidence of a clear clinical benefit of gemcitabine compared with 5-fluorouracil [35]. In that study, it was shown that the 1-year survival rate of pancreatic cancer patients treated with gemcitabine was 18% versus 2% with 5-fluorouracil. Since then, the nucleoside analogue gemcitabine is the first line and the gold standard drug for all stages of advanced pancreatic cancer [4,36]. However, its efficacy is unsatisfactory mainly due to inherent or rapid development of resistance.
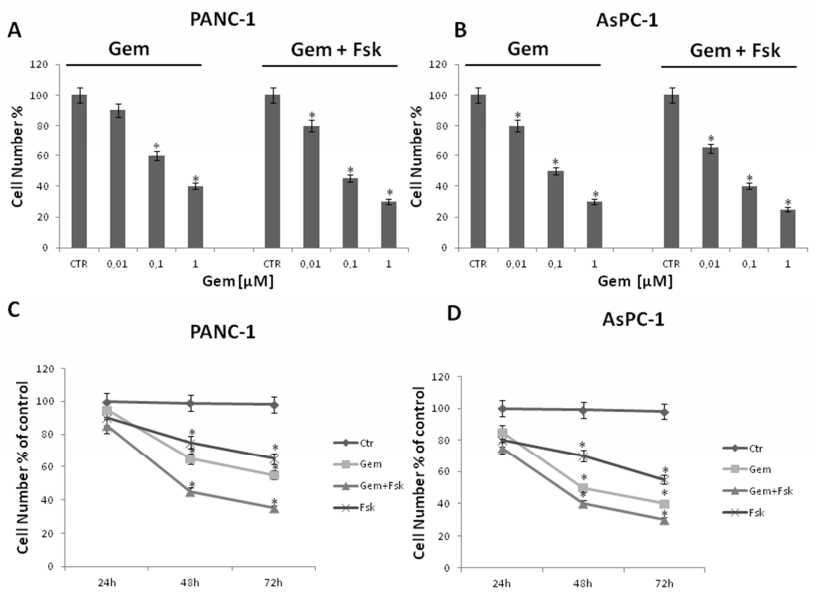
Then, we investigated whether forskolin could enhance the sensitivity to gemcitabine of pancreatic cancer cells. To note, to possibly increase the significance and relevance by our findings, we use low concentrations of gemcitabine (from 0.01 to 1 μM). More in details, we treated or not (control) Panc-1 and AsPC-1 cells with increasing concentrations of gemcitabine (0.01, 0.1, 1 μM), in the presence or absence of 10 µM forskolin for 48 h (Figure 3). After treatments, direct cell number counting as well as conventional tetrazolium-based (MTT) assays were performed.
As expected, proliferation of both Panc-1 and AsPC-1 cells was inhibited by gemcitabine in a dose-dependent manner, although at different extent, with AsPc-1 cells being more sensitive [37,38]. Interestingly, we found that in both pancreatic cancer cell lines, the presence of forskolin strongly enhanced the anti-proliferative effects of gemcitabine in all combinations. Notably, at low dose 0, 1 μM gemcitabine, the growth inhibition in Panc-1 and AsPC-1 cells increases from ≈ 40% to ≈ 55%, and from ≈ 50% to ≈ 60% in the presence of forskolin, respectively (Figure 3A and B). Moreover, we also looked at forskolin/gemcitabine combination-induced antiproliferative effects during a time course. Cells were exposed to no (0 μM, control) or low (0.1 μM) gemcitabine in the presence or absence of 10 µM forskolin for 24, 48 and 72 h (Figure 3C and D). Thereafter, direct cell number counting as well as conventional tetrazolium-based (MTT) assays were performed. As expected, cell proliferation was decreased by gemcitabine in a time-dependent manner. Remarkably, the growth inhibitory effects induced by the forskolin/gemcitabine combination were significantly higher than those caused by gemcitabine alone at every time points. Overall, the above data indicate that forskolin enhances gemcitabine-induced antiproliferative effects in pancreatic cancer cells.
3.4. Forskolin potentiates the gemcitabine-induced antiproliferative effects in pancreatic cancer cells by both inhibiting cell cycle and inducing cell death
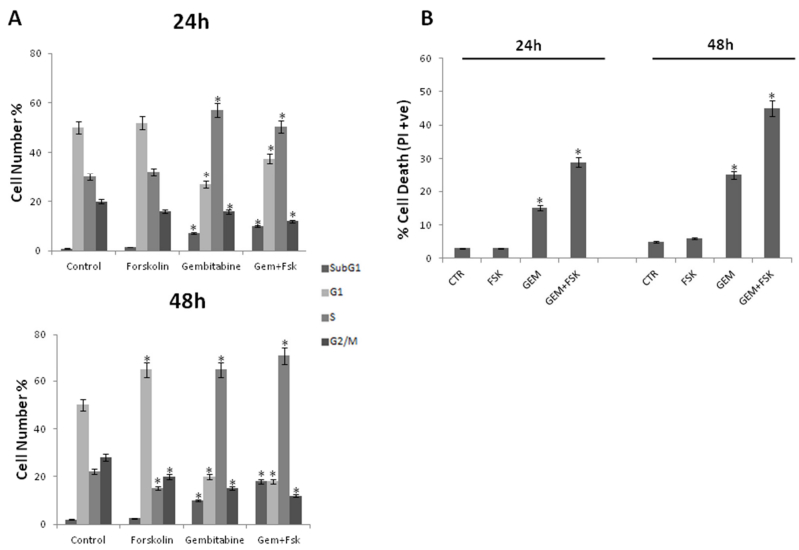
To better understand the enhancement by forskolin of gemcitabine-induced antiproliferative effects, Panc-1 and AsPC-1 cells were exposed or not (control) to 0.1 μM gemcitabine in the presence or absence of 10 µM forskolin for 24 and 48 h. Then, cells were fixed and used for flow cytometry-based assays of cell-cycle progression. Notably, it is known that the deoxycytidine analogue gemcitabine mainly causes cells to accumulate in S-phase [39,40]. Moreover, it is also reported that gemcitabine can differently cause cell cycle modifications and cell death induction in pancreatic cancer cells depending on cell type, cell culture conditions, time and dose of treatment [41,42,43]. Accordingly, Figure 4A shows that gemcitabine-treated cells are differently distributed in the cell cycle phases compared to control untreated ones. Interestingly, in Figure 4A it is also shown that forskolin strongly affects the cell cycle profiles in response to gemcitabine both at 24 and 48 hours. Moreover, a sub-G1 population containing a hypodiploid DNA content (≈ 15%) was observed already at 24 hours and more evident after 48 hours in response to forskolin/gemcitabine combination. Appearance of sub-G1 cells is consistent with cell death. This was further confirmed by measurement of cell death by more sensitive PI uptake assay [26]. In Figure 4B it is clearly shown that forskolin/gemcitabine combination induced a strong time-dependent increase of cell death population, compared to gemcitabine alone, whereas the treatment with forskolin alone did not cause any relevant change in the percentage of cell death (Figure 4, panel B; Figure 2).
Overall, the above data suggest that forskolin potentiates the gemcitabine-induced antiproliferative effects by both affecting cell cycle and inducing cell death.
3.5. Forskolin relevantly affects Erk1/2 and Stat3 phosphorylation levels in response to gemcitabine in pancreatic cancer cells
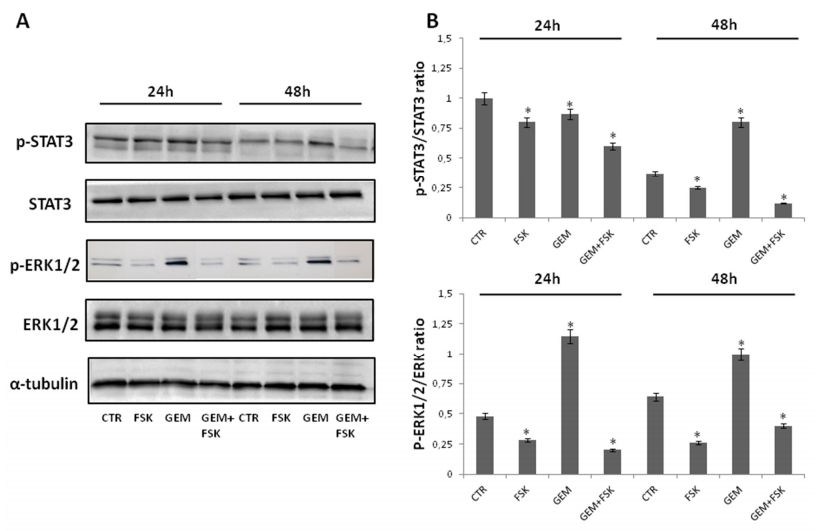
The extracellular signal-regulated kinase (Erk) and signal transducer and activator of transcription 3 (Stat3)-dependent signaling pathways are relevant to pancreatic cancer, as well as to other tumors, and they are frequently activated in [44,45]. Consistently, increasing studies indicate that suppression of Erk and/or Stat3 activation results in the inhibition of proliferative, migratory capabilities and chemosensitivity increase of pancreatic cancer cells [27,28,29,30,46,47,48,49,50,51,52,53,54]. In order to investigate the possible role of Erk1/2 and/or Stat3 in the potentiation by forskolin of gemcitabine-induced antiproliferative activity in pancreatic cancer cells, Western blotting was applied to examine the expression and phosphorylation (activation) of Erk1/2 and Stat3 proteins from extracts of Panc-1 and AsPC-1 cells treated or not (control) with forskolin, gemcitabine and forskolin/gemcitabine combination for 24 and 48 hours (Figure 5). Notably, Figure 5 shows that forskolin/gemcitabine combination induced in Panc-1 cells a strong decrease in both Erk activation and Stat3 activation, evident at both 24 and 48 hours, without affecting significantly the total amount of proteins examined. Similar results were obtained also in AsPC-1 cells (data not shown).
Consistent with the antiproliferative action of forskolin and previous similar findings [34], overall the above data indicate that forskolin treatment of pancreatic cancer cells interferes with the modulation of Erk1/2 and Stat3 activation in response to gemcitabine and suggest that the forskolin-mediated inhibition of Erk and Stat3 signaling is involved in the enhancement of sensitivity to gemcitabine by forskolin of pancreatic cancer cells.
4. Discussion
Pancreatic cancer is one of the most quickly fatal human cancers with 5-year survival rates remaining constant at less than 10% [1,2]. Gemcitabine is the first line and the gold standard drug for pancreatic cancer till now. However, its efficacy is unsatisfactory and provides only modest survival benefit [3,4,35]. Therefore, identification of compounds within novel combinatorial approaches that improve gemcitabine activity to possibly increase the survival of pancreatic cancer patients are warranted and consistently being investigated [27,28,30,43,48,49,51,52,53,55,56,57].
Forskolin is emerging as one of the most interesting natural compounds from medicinal plants to possibly use in cancer therapy [9]. Recently, the cAMP elevating agent forskolin has been shown to enhance the sensitivity to conventional antineoplastic drugs in myeloma and in colorectal cancer cells [17,21]. Importantly, here we describe that the natural compound forskolin has by itself profound antiproliferative and antimigratory effects against human pancreatic cancer cells, in combinatorial treatments markedly potentiates the antiproliferative effect of the chemotherapeutic agent gemcitabine, by both cell cycle inhibition and cell death induction, preceded and accompanied by the inhibition of Stat3 and Erk1/2 signaling pathways.
The complexity of the cellular signaling network in pancreatic cancer cells opens the opportunity to combined inhibition of multiple pro-cancer pathways, which could result in superior anti-cancer activity. Stat3 belongs to a family of latent cytoplasmic transcription factors that are activated by phosphorylation of a critical tyrosine residue, dimerization and nuclear translocation, upon binding of cytokines and growth factors to their membrane receptors [58]. Multiple lines of evidence place Stat3 at a central node in the development, progression, and maintenance of many human tumors based on its ability to transactivate a number of genes whose products are involved in cell proliferation and survival, invasion, metastasis and angiogenesis [59]. Constitutive activation of Stat3 is a frequent occurrence in various tumors, including pancreatic cancer, and has been demonstrated in a number of pancreatic cancer cell lines [27,29,45,54,60]. In addition, high levels of Stat3 activity have been associated with poor outcomes and have also been found to predict intrinsic drug resistance to chemotherapy and Stat3 inhibition has been evaluated as a possible strategy to increase tumor cell response to cytotoxic agents [60,61,62]. Thus, Stat3 has been validated as an anticancer target in several contexts [63,64].
At the meantime, over-activation of Ras/Mek/Erk pathway is observed in over 90% of pancreatic cancer patients, which is known as a major contributor of pancreatic cancer progression and resistance [44,65,66]. Very interestingly, in the current study, we show that the forskolin treatment of pancreatic cancer cells affects the modulation of Erk1/2 and Stat3 activation in response to gemcitabine, strongly suggesting that the forskolin-mediated inhibition of Erk and Stat3 signaling is involved in the enhancement of sensitivity to gemcitabine. Notably, our results are in agreement with previous findings indicating that forskolin can exert anticancer activities by inhibiting Erk1/2 and Stat3 signaling pathways [18,34,67,68]. However, we do know that the detailed molecular mechanisms underlying the forskolin-induced enhancement of sensitivity to gemcitabine in pancreatic cancer remain unclear and need to be investigated further. By the way, whatever the exact mechanism(s) here we report that exogenous addition of the natural occurring molecule forskolin inhibits the proliferative and survival Erk1/2 and Stat3 signaling pathways and acts as a potent gemcitabine sensitizer agent in pancreatic cancer cells.
Currently, gemcitabine is only approved single chemotherapeutic drug for the clinical treatment of pancreatic cancer; however, its activity in increasing patients' survival is very far from being ideal [3,4,36,55]. As a consequence, gemcitabine sensitizers are actively being searched from many scientific groups [27,28,30,43,48,49,51,52,53,54,55,56,57]. Importantly, our results confirm and extent the antiproliferative and antimigratory action by forskolin on pancreatic cancer cells enforcing the evidence of forskolin as a natural compound with anticancer potential by itself and demonstrate that co-administration of forskolin significantly increases gemcitabine's anti-pancreatic cancer activity in vitro. Remarkably, forskolin is one of the commercially important herbal ingredients for weight loss dietary supplements in the global market [10]. Its favorable effects on body fat management have been clearly reported and clinical studies with oral administration of forskolin for up to 50 mg/day as a weight loss agent have not shown relevant side effects in patients [11,12,13]. By our findings further studies exploring forskolin as a gemcitabine sensitizer/adjuvant in vivo might be strongly encouraged.
5. Conclusions
The cAMP natural elevating agent forskolin has been used for centuries in traditional medicine, is considered affordable and its safety has also been documented in conventional modern medicine.
Our findings that forskolin has by itself profound antiproliferative and antimigratory effects against human pancreatic cancer cells, inhibits gemcitabine-induced activation of Stat3 and Erk1/2 signaling pathways and increases the sensitivity to gemcitabine of pancreatic cancer cells, enforce the evidence highlighting the potential for its possible use in the development of novel and effective modalities for therapeutic intervention in pancreatic cancer.
Acknowledgments
This study was supported by funds from Ministry of Education, University and Scientific Research (PRIN) and from University of Campania "Luigi Vanvitelli".
Conflicts of Interest
The authors declare no conflict of interest.








 DownLoad:
DownLoad: